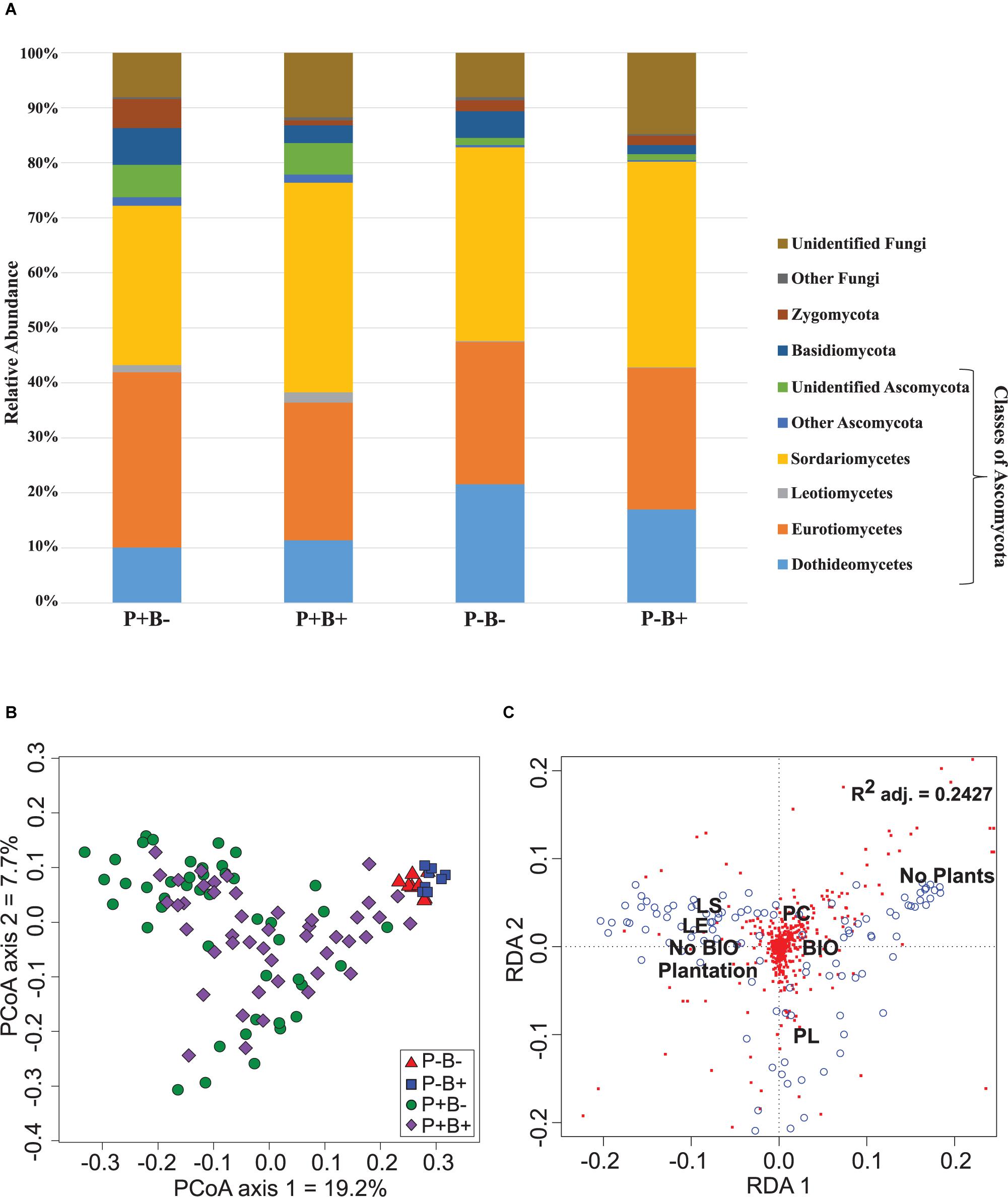
In this role, he explores needs for visual and statistical discovery software in the clinical segment of the pharmaceutical and healthcare industries. JMP® Genomics is a statistical discovery software package for researchers that enables statistical geneticists, biologists, bioinformatics experts, and statisticians to uncover meaningful patterns in high-throughput genetics, expression microarray, and proteomics data. JMP Product Manager, SAS Geoffrey Mann is JMP Product Manager for the health and life sciences industry. It links advanced statistics and graphics, creates reports from CDISC data, and helps reviewers migrate into the modern review environment. Praise for the Fourth Edition 'As with previous editions, the authors have produced a leading textbook on regression. MEDICAL MONITORS SAFETY AND EFFICACY The goal of a medical monitor is to evaluate the safety (adverse. JMP® Clinical shortens the drug development process by streamlining safety reviews during clinical trials. JMP® Pro is the advanced analytics version of JMP, providing all the tools for visual data access and manipulation, interactivity, comprehensive analyses, and extensibility found in JMP. JMP® Live helps you share JMP analyses in an online space designed for interactive collaboration in your organization. It helps you see and explore data, spot relationships and trends, and dig into areas of interest. Accelerated new product introductions through knowledge re-use.Īs the pharmaceutical industry transitions into a more metrics based future, there has never been a better time to invest in realizing the enormous benefits of using data-driven analytics.Designed for dynamic data visualization and analytics on the desktop, JMP enables data access and processing statistical analysis design of experiments multivariate analysis quality and reliability analysis scripting graphing and charting.Managing pipeline risk with increased understanding.Uncovering issues unidentifiable without central statistical monitoring methods.Reducing the frequency of interactions with regulatory agencies.Identifying the causes of data quality issues faster.Saving up to 30 percent of the costs of running trials.The potential benefits of these cutting-edge methodologies are: Using three examples, he will show how the informed application of statistically designed experiments, one of the cornerstones of QbD, can dramatically increase your clinical, product and process understanding for the same use of resources. Ron Kenett, Chairman of the KPA Group, insights through analytics, will provide an overview of the data-driven aspects of Quality by Design (QbD) and the associated FDA guidance. Data quality and fraud detection methods to identify outlying patients and sites in a clinical trial.
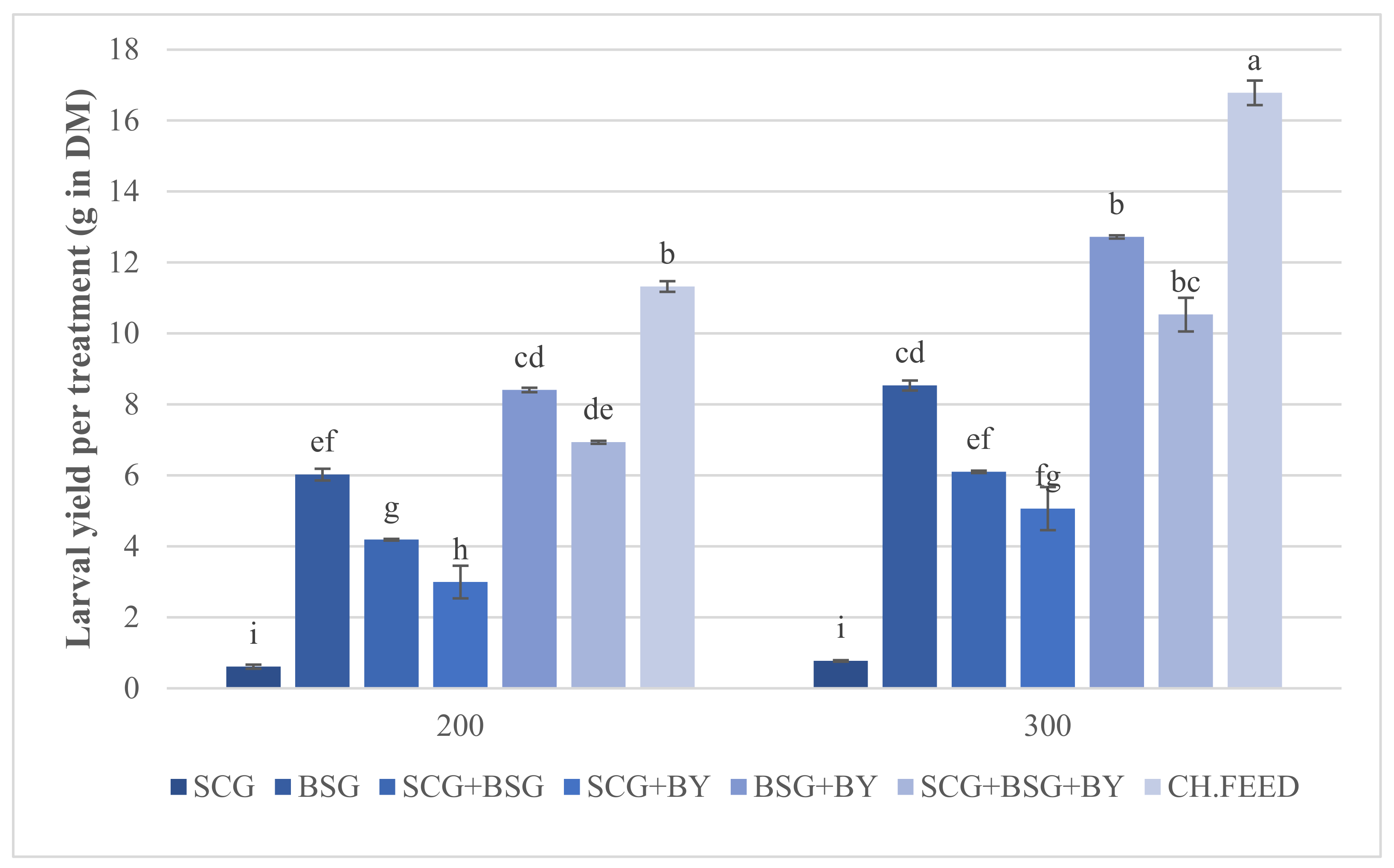
Central statistical monitoring methods.Risk-based monitoring that aligns to risk indicators, thresholds and actions set forth by the TransCelerate methodology documents.

Geoffrey sas jmp code#
The practitioner's primary taxonomy code is 1223E0200X with license number 060421 (NY). The NPI number of Geoffrey Sas is 1407406218 and was assigned on September 2019. Using these methods will improve and align your monitoring process with recent guidance documents created by the FDA and EMA. Geoffrey Sas is a provider established in New York, New York and his medical specialization is dentist (endodontics). Previously, Geoffrey was a Product Manager, Jmp, Health & Life Sciences at SAS. Geoffrey Mann is a Research & Development Manager, Advanced Analytics at JMP based in Cary, North Carolina. JMP Genomics analyzes microarray, genetic marker and proteomics data.
Geoffrey sas jmp how to#
Geoffrey Mann, JMP Product Manager at SAS, will discuss how to modernize your on-site clinical trial monitoring by using the latest methods in risk-based monitoring, central statistical monitoring and clinical oversight monitoring. Research & Development Manager, Advanced Analytics at JMP. I am involved in both JMP Genomics and SAS Drug Development. Similarly, whilst the WHO draft guidance (Sept 2015) contains requirements on queries that allude to training, they can only really be addressed by using analytics. The addendum to ICH E.6 (GCP) brings us QMS, RBA, quality tolerance limits and quality reporting. The JMP family of products provide many capabilities that help you to leverage data better, and this seminar focuses on two distinct but related areas of application.Īndrew Lawton, CEO of Risk Based Approach Ltd, will examine and interpret forthcoming regulations and guidance changes and the use of big data and analytics. In the pharmaceutical industry there is no guidance which states that you must use data-driven analytics, yet this is the only effective way to meet the requirements imposed by regulatory agencies.
